The density maps and coordinate files will be available upon publication.įunding: KW is supported by post-doc scholarships from The Independent Research Fund Denmark and The Lundbeck Foundation. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.ĭata Availability: The cryo-EM density maps and the coordinate (pdb) files generated in this study have been deposited to the Electron Microscopy Data Bank and Protein Data Bank (PDB), respectively, and with the following accession codes: EMD-4645 and 6QV6 for map 0 at pH 7.5 EMD-4647 and 6QVC for map 1, pH=7.5 EMD-4649 and 6QVD for map 2, pH=7.5 EMD-4646 and 6QVB for map 3, pH=7.5 and EMD-4657 and 6QVU for map, pH=6.2). Received: OctoAccepted: MaPublished: April 25, 2019Ĭopyright: © 2019 Wang et al. Lieberman, Georgia Institute of Technology, UNITED STATES PLoS Biol 17(4):Īcademic Editor: Raquel L. (2019) Structure of the human ClC-1 chloride channel. Our results help with understanding the principal determinants that govern CLC proteins and may guide downstream translational applications to combat muscle pathologies.Ĭitation: Wang K, Preisler SS, Zhang L, Cui Y, Missel JW, Grønberg C, et al. Our data suggest how ClC-1 is regulated by environmental cues to allow opening and closure, thereby permitting attenuation of muscle function. Here, we have analyzed the structure of human ClC-1 and revealed the high similarity of its ion conducting pathway to those observed in other CLC members, including prokaryotic and algal transporters. The CLC channel ClC-1 is critical to skeletal muscle excitability and has been proposed as a target to alleviate neuromuscular disorders. Structurally, CLCs form dimers possessing a separate ion translocation pathway in each monomer, and they can operate as either channels or transporters that exchange chloride for protons. The structure also provides a framework for analysis of mutations causing myotonia congenita and reveals a striking correlation between mutated residues and the phenotypic effect on voltage gating, opening avenues for rational design of therapies against ClC-1–related diseases.Ĭhloride transporting CLC proteins are expressed in a wide range of organisms, and the family encompasses several members with numerous roles in human health and disease by allowing movement of chloride ions across the membranes that encapsulate cells and cellular organelles. Comparison of structures derived from protein studied in different experimental conditions supports the notion that pH and adenine nucleotides regulate ClC-1 through interactions between the so-called cystathionine-β-synthase (CBS) domains and the intracellular vestibule (“slow gating”). These characteristics agree with the lower chloride flux of ClC-1 compared with ClC-K and enable us to propose a model for chloride passage in voltage-dependent CLC channels. 
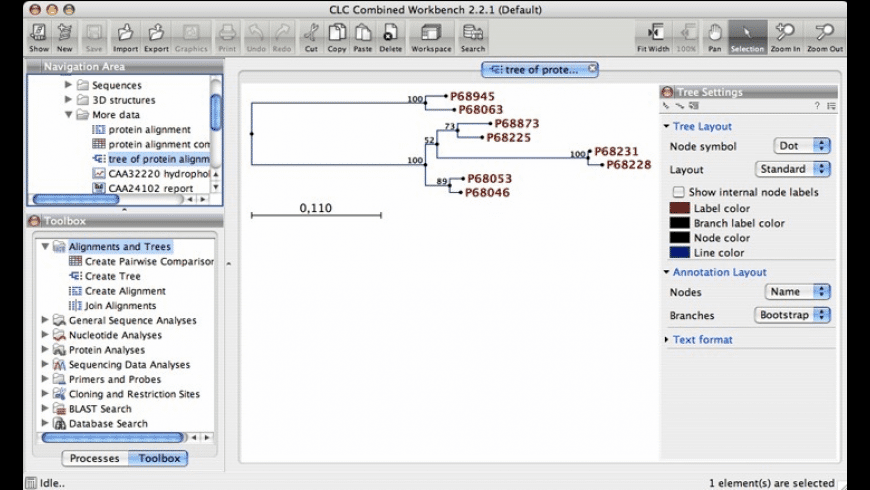
The chloride conducting pathway exhibits distinct features, including a central glutamate residue (“fast gate”) known to confer voltage-dependence (a mechanistic feature not present in ClC-K), linked to a somewhat rearranged central tyrosine and a narrower aperture of the pore toward the extracellular vestibule. Here, we present the cryo-electron microscopy (cryo-EM) structure of human ClC-1, uncovering an architecture reminiscent of that of bovine ClC-K and CLC transporters. 
Malfunction of ClC-1 is associated with myotonia congenita, a disease impairing muscle relaxation. ClC-1 protein channels facilitate rapid passage of chloride ions across cellular membranes, thereby orchestrating skeletal muscle excitability. 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
